Welcome to TAmiRNA’s resource hub!
Access key background, protocols, scientific posters, and practical tips to get the most out of our products and services — from microRNA analysis to sequencing and bioinformatics.
Stay tuned for updates and boost your research with our latest insights.

White Paper
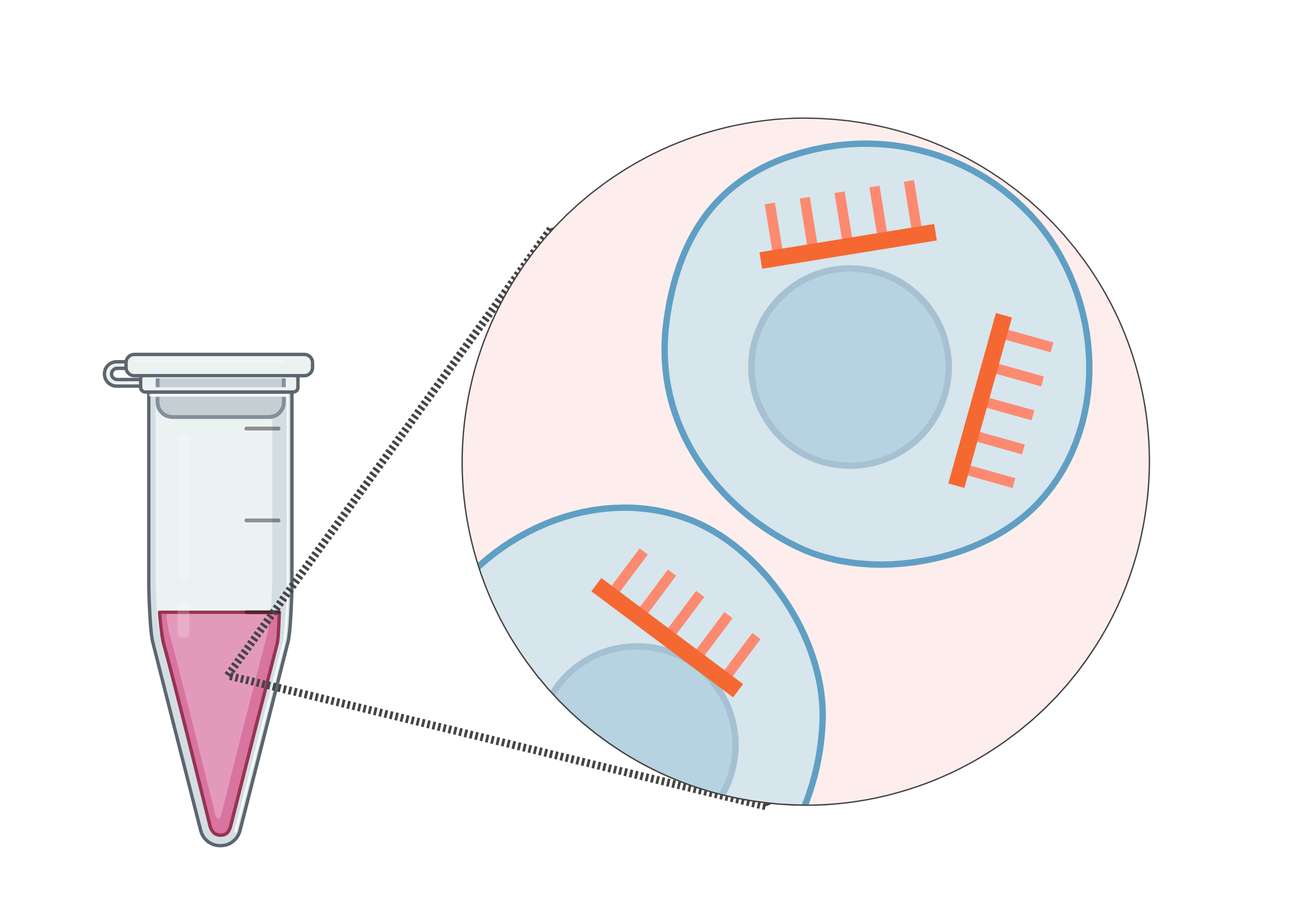
Defining Potency-Relevant & Tissue-Specific microRNA Cargo in RoosterBio hMSC-Exosomes Characterized Using TAmiRNA’s miND® Workflow
January 2026
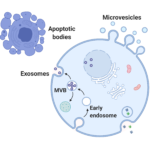
Tech note 10
Extracellular vesicles (EVs) as a novel source for biomarker development – analysis pitfalls and important considerations.
August 2021
Tech note 09
Determining the limit of detection of TAmiRNA’s SARS-CoV-2 IgM/IgG Antibody Rapid Test using the WHO International Standard NIBSC 20/136.
Juli 2021

Tech note 08 (Deutsch)
Nachweis der Immunantwort nach SARS-CoV-2-Impfung mit Vaxzevria (COVID-19-Impfstoff AstraZeneca) mit Hilfe des TAmiRNA SARS-CoV-2 IgM/IgG Antikörper-Schnelltest.
Juli 2021

Tech note 08 (English)
Verification of immune response after SARS-CoV-2 vaccination with Vaxzevria (COVID-19 Vaccine AstraZeneca) using TAmiRNA SARS-CoV-2 IgM/IgG Antibody Rapid Test.
Juli 2021

Tech note 07 (Deutsch)
Überprüfung der Immunantwort nach SARS-CoV-2 Impfung mit Pfizer/BionTech (BNT162) mit Hilfe des TAmiRNA SARS-CoV-2 IgM/IgG Antikörper Schnelltests.
März 2021

Tech note 07 (English)
Verification of the immune response after vaccination with Pfizer/BioNTech (BNT162) using TAmiRNA SARS-CoV-2 IgM/IgG Antibody Rapid Test.
March 2021

Tech note 05
Quality controls and best practices for analyzing microRNAs in cell-free biofluids by RT-qPCR.
April 2019