news
New publication in Biomaterials
Huge congrats to Cansu Gorgun and the teams at Royal College of Surgeons in Ireland (RCSI) and Trinity College Dublin on their new publication in Biomaterials! This study is a fascinating look at how we can "train" macrophages to do two things at once. By reprogramming their metabolism, the team produced hybrid M1/M2 extracellular vesicles — a "dual-action" package that combines pro-inflammatory and [...]
TAmiRNA @ RNA Leaders Europe Conference
Vienna, Austria – 17.-19. March 2026 TAmiRNA is pleased to announce that CEO/CSO Matthias Hackl and Head of R&D Teresa Krammer will attend the upcoming RNA Leaders Europe Conference, one of the leading industry events dedicated to RNA therapeutics, innovation, and translational research. RNA Leaders Europe brings together key stakeholders from academia, biotech, pharma, and technology providers to discuss the latest scientific advances, [...]
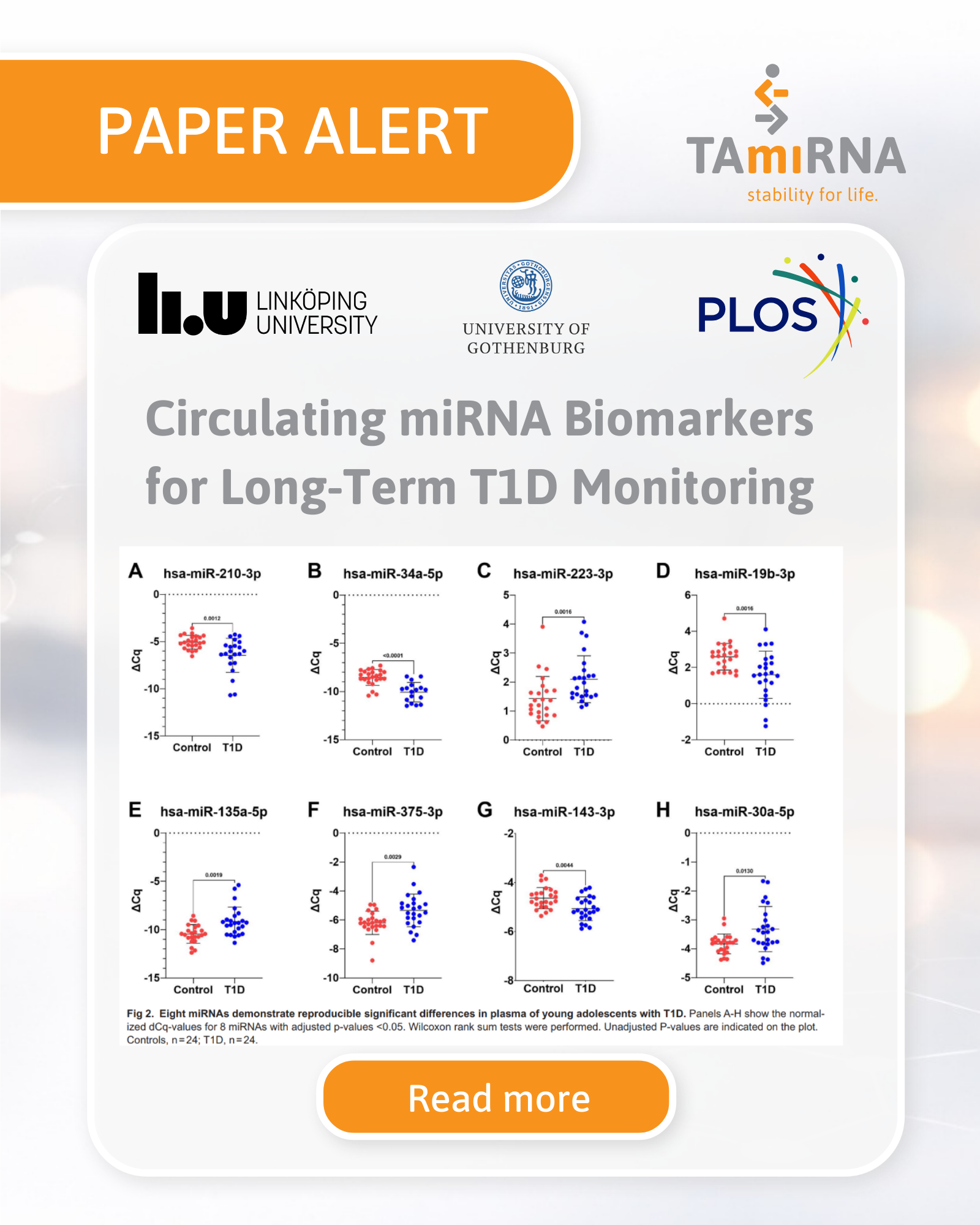
Independent Validation of Circulating microRNAs as Biomarkers in Type 1 Diabetes
TAmiRNA is proud to contribute to a newly published peer-reviewed study demonstrating the independent validation of circulating microRNAs (miRNAs) as biomarkers in adolescents with long-standing type 1 diabetes (T1D). The study, published in PLOS ONE (2026), strengthens growing evidence that circulating microRNAs serve as minimally invasive biomarkers with potential applications in disease monitoring and precision medicine. Study Overview: microRNA Biomarkers in Adolescent Type [...]
New white paper with RoosterBio on omics-tools for EV characterization
In this white paper, TAmiRNA and RoosterBio present a comprehensive small RNA-sequencing–based characterization of mesenchymal stem cell (MSC)–derived exosomes produced using RoosterBio’s standardized, xeno-free manufacturing platform. The study was designed to assess both the analytical robustness of TAmiRNA’s miND® small RNA-sequencing workflow and the biological relevance of microRNA cargo profiling as an advanced “omics-based” characterization strategy for therapeutic extracellular vesicles (EVs). Using technical [...]
TAmiRNA Research on microRNA Biomarkers for Periprosthetic Joint Infections Featured on Wien Heute, One of Austria’s Most-Watched Regional News Programs
Research conducted by TAmiRNA in collaboration with the Michael Ogon Laboratory (Orthopädisches Spital Speising) on microRNA-based biomarkers for the early detection of periprosthetic joint infections (PJI) was recently featured on Wien Heute, one of Austria’s most widely viewed regional television news programs (ORF, January 3, 2026). The coverage highlights both the clinical relevance of the problem and the translational potential of novel molecular [...]
Synovial Fluid and Serum microRNA Signatures in Equine Osteoarthritis
New research highlights the potential of circulating miRNAs as biomarkers for joint degeneration A new peer-reviewed study has been published in the International Journal of Molecular Sciences (IJMS): “Synovial Fluid and Serum MicroRNA Signatures in Equine Osteoarthritis.” This work provides important insights into how microRNAs (miRNAs) in serum and synovial fluid reflect the presence of osteoarthritis (OA) in horses. 🎯 Study Aim The [...]
TAmiRNA and Orthopaedic Hospital Speising Publish Landmark Data on promiR® for Accurate Diagnosis of Periprosthetic Joint Infections
On November 19th, 2025, the study “Synovial Fluid MicroRNA Biomarkers Enable Accurate Diagnosis of Hip and Knee Periprosthetic Joint Infections” by Frank et al. was published in the Journal of Bone and Joint Surgery. This peer-reviewed publication marks an important milestone in TAmiRNA’s biomarker development program and presents promiR®, our novel microRNA-based diagnostic panel designed to improve the pre-operative assessment of [...]
Distinct microRNA profiles in human amniotic membrane its vesicular and non-vesicular secretome revealed
Vienna, Austria – November 2025 TAmiRNA scientist have contributed significantly to a new study published by the Ludwig Boltzmann Institute for Traumatology, which is titled “Distinct miRNA profiles in human amniotic tissue and its vesicular and non-vesicular secretome”. The article was published in Frontiers in Cell and Developmental Biology on October 29th 2025. Human amniotic membrane (hAM) has been used in tissue regeneration [...]
TAmiRNA launches the new lyophilized miND® spike-in kit for small RNA-sequencing quality control
Vienna, Austria – November 2025 TAmiRNA proudly announces the release of its miND® spike-in kit in a new lyophilized format, optimized for the preparation and analysis of 96 small RNA-sequencing libraries. This upgraded format improves handling, stability, and integration with standard small RNA-sequencing workflows, ensuring seamless compatibility with commonly used library preparation kits. The new lyophilized miND® spike-in kit combines ease of use [...]
TAmiRNA @ Precision Medicine EV Forum
TAmiRNA to Exhibit at the Precision Medicine EV Forum 2025 in Cambridge Vienna, October 2025 – TAmiRNA GmbH, a leading provider of extracellular vesicle (EV) transcriptomic services, is pleased to announce its participation as an exhibitor at the Precision Medicine EV Forum 2025, taking place in Cambridge, UK, from October 27th to 28th. The forum, a key event focused on the translational aspects [...]